OxySoft
Displays relative concentration changes in oxy-, deoxy-, total hemoglobin, and tissue saturation index (TSI) in real-time
Compatible with all our NIRS devices
Real-time calculation of oxy-, deoxy-, total hemoglobin concentration changes, and tissue saturation index (TSI)
Displays easy-to-understand 2D and 3D plots
Supports simultaneous recordings of multiple devices
Records and generates events from a wide range of sources
All standard analysis techniques can be applied in real-time or posthoc
Compatible with e.g. HOMER, NIRS-SPM, FieldTrip, NIRStorm, Polhemus, and more
Exports raw data in various formats
Enables real-time streaming of data to e.g. Matlab or Python via Lab Streaming Layer (LSL)
The software
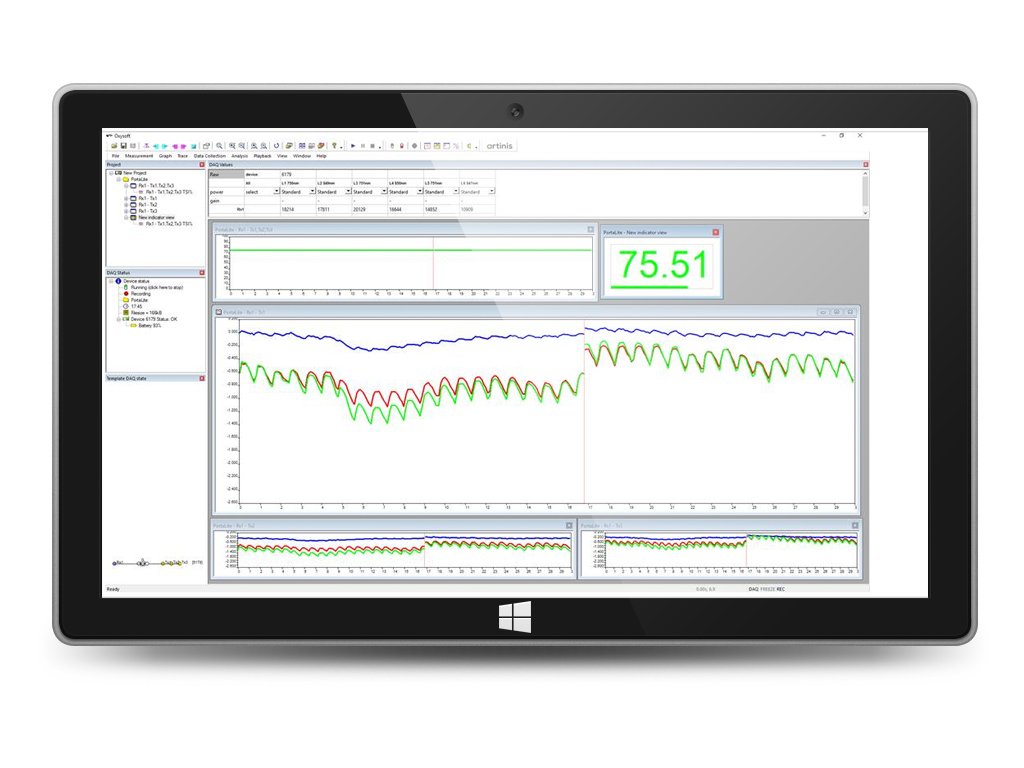
OxySoft is our proprietary NIRS recording and analysis software. OxySoft allows robust recordings even of multiple devices at the same time, in any thinkable combination. This means you can combine recordings from several OxyMon’s, PortaMon’s, PortaLite’s, OctaMon’s, or the Brite Family devices within one measurement by using just one program. All data is synchronized and stored in one data file. You can analyze your data in real-time, during the recording, or afterwards.
Features
-
OxySoft is a dedicated software application used to collect, store, view, and analyse all necessary data from the Brite Family devices, PortaMon, PortaLite, OctaMon and OxyMon. This user-friendly software is developed by Artinis Medical Systems and highly customisable to individual requirements.
-
Data from OxySoft can be analysed in a variety of ways. First, you can analyse data within OxySoft. OxySoft offers a variety of simple standard analysis techniques, such as filtering, averaging and statistical analysis. Second, data can also be exported to standard data formats, such as .csv, .txt or .xls file format. Third, we provide a MATLAB script, which allows importing your data obtained from Oxysoft directly into several MATLAB toolboxes such as Homer2, NIRS-SPM or FieldTrip. The script also allows importing the raw data, so that you are free to do any preferred analysis from within MATLAB.
-
Main features include:
1. Real-time data collection2. Connection and data collection of multiple Artinis devices at the same time to guarantee perfect synchronization
3. Creation of projects to store related files and set-up analysis for all files simultaneously
4. Generation of events using various methods, e.g. keyboard or external inputs
5. Application of various types of filters
6. Re-sampling of data
7. Calculation of average, mean, standard deviation and regression
8. Calculation of differences between selected timeframes
9. Calculation of block average, including detrend function
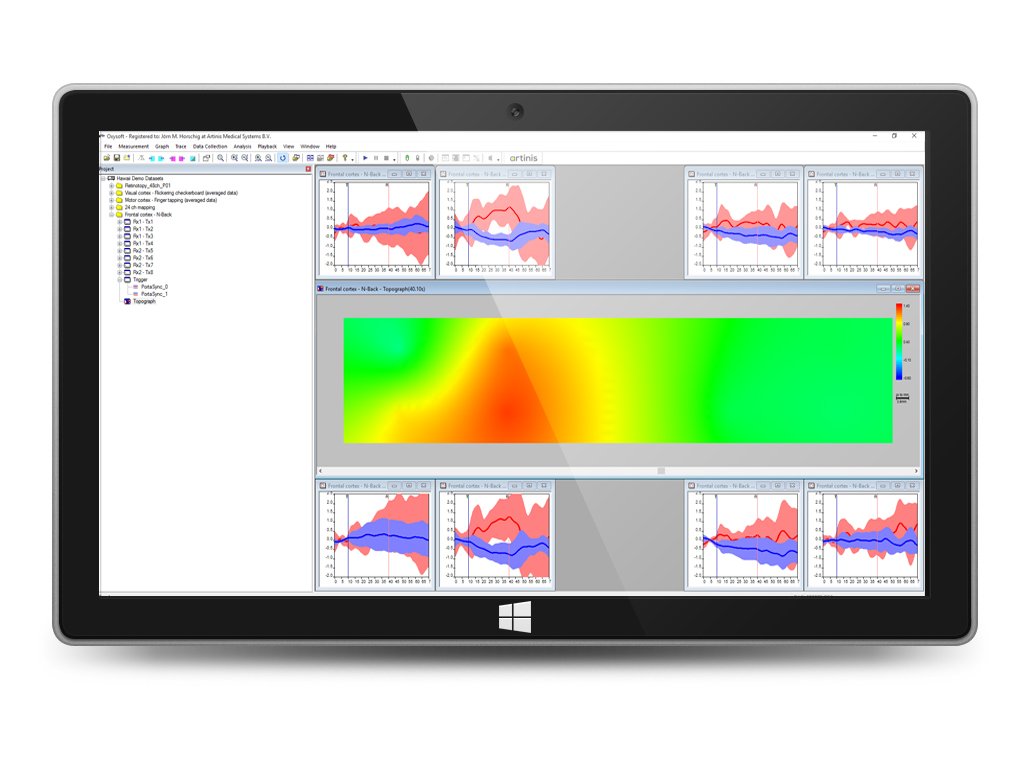
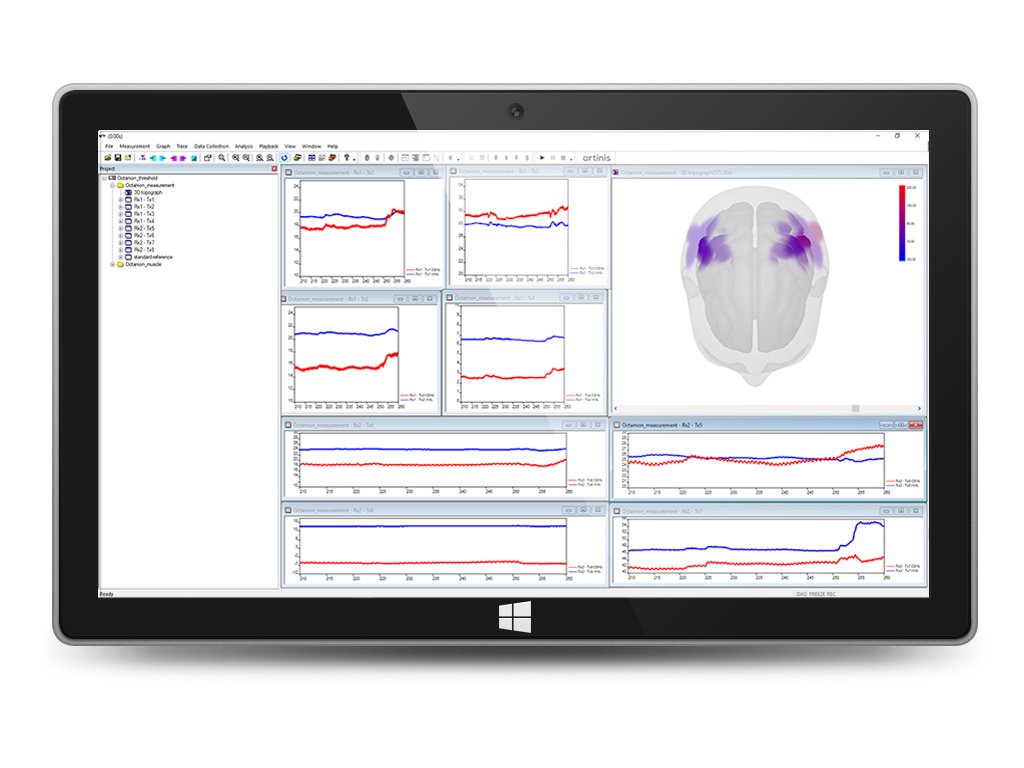
10. Creation of two- of three-dimensional topo graphs to display (brain) activity for brain mapping
11. Creation of spectrum graphs
12. Comparison between traces
13. Export of data files to different formats (for instance text, Excel, XML)
14. Real-time data in- and export from / to Matlab and other software using LSL
15. Offline data export to Matlab and Matlab based (SPM) software packages
16. Import of data files collected with Artinis devices and other instruments
New in OxySoft 4.0:
17. Export of data files directly to .snirf format
18. Performance of device event mapping
19. Retrieval of lost data due to Bluetooth connection issues using the recovery tool
20. Submission of feedback directly due to software
OxySoft 4
OxySoft 4, our elevated software version, comes with exciting features to facilitate the acquisition and analysis of NIRS and fNIRS data and provides maximum flexibility. It incorporates:
Direct and easy export to .snirf
Retrieve data integrity in case of a lost Bluetooth connection using the recovery tool
Direct Creation of events using device event mapping
Intuitive signal quality assessment with improved UI and implementation of signal quality index (SQI)
Options to directly provide feedback and get support with a few clicks
The OxySoft 4 is compatible with all our latest devices, e.g. Brite family, PortaLite MKII, and PortaSync.
OxySoft extensions
We offer the opportunity to access extended visualization options, designed to optimize your measurement protocol, better review your results, and to optimally support your high-impact neuroscience publication. The OxySoft 3D extension can visualize your data on 3D models of the brain of the human head based on the widely used MNI templates.
With the 3D extension you are enabled to:
Visualize your data on 3D models of the brain of the human head based on the widely used MNI templates.
Use a built-in, intuitive user interface for the Polhemus fast-track device
Automatically store the digitized positions of the optodes
Oxysoft 3D & Polhemus digitizer
We have partnered with Polhemus Inc., which creates devices for digitizing objects such as the Polhemus Patriot or Polhemus Fastrack device. Using these devices you can precisely measure the position of the optodes on the subject’s head. OxySoft 3D guides you through this process with a built-in, intuitive user interface. No third-party software is needed for this process. Alternatively, you can import digitized positions from other manufacturers such as ANT’s Xensor or Magstim’s EEG pinpoint device. OxySoft automatically stores the digitized positions of the optodes and transforms and then visualizes them on the integrated 3D models. This not only allows you to better review your data and results but additionally allows you to report back the optode positions in MNI coordinate space.
The quick start guide
We are happy to guide your research journey with the OxySoft quick start guide video series. This OxySoft tutorial video series is now available on Artinis YouTube channel. You will find the a to z whereabouts of OxySoft NIRS recording and analysis software in one playlist.
In addition, you will also get insightful tips & tricks coming right from our specialists.
Artinis Medical Systems BV
Compatible analysis toolbox
The MediBrite is a CE marked medical device according to Medical Device Regulation (EU) 2017/745.
Other Artinis NIRS products are intended to be used for research applications only. Other Artinis products are not sold as Medical Device as defined in Medical Device Regulation (EU) 2017/745. Our products are not designed or intended to be used for the diagnosis or treatment of disease.







We are proud of the enhanced performance of our recently released NIRS monitoring software. In this blog post, we explain, which of our devices are supported in OxySoft 4.0 and higher.